White and red blood, as well as platelets are derived from haematopoietic stem cells, which live in the bone marrow cavity. People affected by leukaemia present with symptoms arising due to an uncontrolled proliferation of white blood cells which are arrested at different maturation stages depending on the type of leukaemia. This abnormality interferes with the balance between growth and development of normal blood cells.
Our daily work at the Krause Lab is based on the understanding of the molecular and cellular interactions between leukaemic cells and the bone marrow microenvironment.
Our challenge is to design novel pharmacological agents which will target the interactions between bone marrow niche cells or other factors and the leukaemia cells at a defined location, but ideally without destroying the physiological cellular compartment and at the same time helping the normal haematopoietic stem cells system to overcome the disease.
In order to answer these questions, we use knock out mice, i.e. mice lacking a particular gene of interest, and we follow up by studying the phenotypic and molecular differences between these leukaemic cells and those not lacking this particular gene, as well as their interactions with the surrounding microenvironment. These models help us understand which of the thousands of components are the factors supporting the development of leukaemia.
We use genetic, physiological and biochemical approaches to answer these questions because it is assumed that certain proteins from the bone marrow microenvironment are able to instruct leukaemic cells to be more aggressive. The mechanism by which a certain protein provides a message to leukaemia cells varies widely between leukaemia type and from patient to patient. We consider this aspect to be of great importance, in order to be able to apply our research to potential personalised treatment strategies. For this reason, we use various molecular techniques that help us predict and further recognize the “postman” responsible for delivering the message to leukaemia cells, while, consequentially, trying to block these messages.
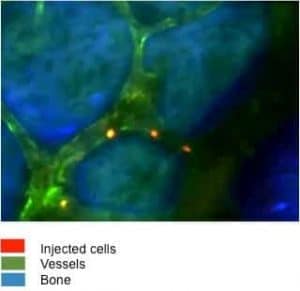
The visualization of these particular cells of interest, as well as their interactions with other cells, confirms our findings and completes this complex picture. In vivo microscopy also has the advantage of helping us discover the reason behind why leukaemia cells have a unique localisation in the bone marrow, thus indicating biological differences and possible approaches to target these differences.
For example, we are interested in characterizing the interaction between leukaemia cells and blood vessels inside the bone. Can we have an analogue of the “Street view-Google map” system in a solid tissue such as bone? Yes. We are able to use a novel and highly potent microscopic technique, whereby we label our cells with a dye and later directly see by eye, when and for how long this injected cell likes to “sit” and “chat” with the blood vessel in a mouse, which may be lacking a certain gene of interest. As mentioned previously, the bone microenvironment communicates with leukaemia cells and orchestrates the cells' behaviour and subsequently the clinical outcome.
This is not the last action and fantasy Hollywood movie but is the scenario of how leukaemic cells find their way home in order to make the disease progress.
The traditional treatment for leukaemia has been to target the leukaemia cells themselves. However, our research is showing that leukaemia cell behaviour heavily depends on the surrounding microenvironment. Thus, it is pivotal to understand how targeting the bone marrow microenvironment itself may add to the eradication of leukaemia stem cells, which is considered a prerequisite for cure.
We strongly advise you to talk with a health care professional about specific medical conditions and treatments.
The information on our site is meant to be helpful and educational but is not a substitute for medical advice.
This is my story of surviving cancer I was diagnosed with Non Hodgins Lymphoma back in 2006 my dad had died of the same marginal within 17th months which is unfortunately the normal. By 2007 I was told I had six months and treatment gave me 6 months more. The in 2008 I was told I had […]
Discover Essential Tips To Craft A Nutritious Diet Plan For Cancer Patients, Manage Side Effects, And Support Overall Health.
"Since My Diagnosis I Have Had Many Conversations With Men About Their Risk, The Need To Be Screened And Prostate Cancer Survival".